Trích:
|
Nguyên văn bởi Dinh Tien Dung
Tại sao nhiệt độ sôi và nhiệt độ nóng chảy của xicloankan lại cao hơn so với ankan tương ứng(xét cùng số nguyên tử C).
|
Boiling point
Alkanes experience inter-molecular van der Waals forces. Stronger inter-molecular the van der Waals forces give rise to greater boiling points of alkanes.
There are two determinants for the strength of the van der Waals forces:
* the number of electrons surrounding the molecule, which increase with the alkane's molecular weight
* the surface area of the molecule
Under standard conditions, from CH4 to C4H10 alkanes are gaseous; from C5H12 to C17H36 they are liquids; and after C18H38 they are solids. As the boiling point of alkanes is primarily determined by weight, it should not be a surprise that the boiling point has almost a linear relationship with the size (molecular weight) of the molecule. As a rule of thumb, the boiling point rises 20 - 30 °C for each carbon added to the chain; this rule applies to other homologous series.
A straight-chain alkane will have a boiling point higher than a branched-chain alkane due to the greater surface area in contact, thus the greater van der Waals forces, between adjacent molecules. For example, compare isobutane and n-butane which boil at -12 and 0 °C, and 2,2-dimethylbutane and 2,3-dimethylbutane which boil at 50 and 58 °C respectively. For the latter case, two molecules 2,3-dimethylbutane can "lock" into each other better than the cross-shaped 2,2-dimethylbutane, hence the greater van der Waals forces.
On the other hand, cycloalkanes tend to have higher boiling points than their linear counterparts due to the locked conformations of the molecules which give a plane of intermolecular contact
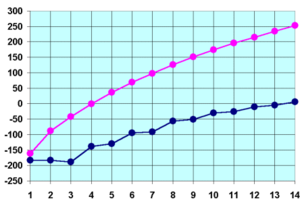
Melting (blue) and boiling (pink) points of the first 14 n-alkanes in °C.
Melting point
The melting points of the alkanes follow a similar trend to boiling points for the same reason as outlined above. That is, (all other things being equal) the larger the molecule the higher the melting point. There is one significant difference between boiling points and melting points. Solids have more ridged and fixed structure than liquids. This rigid structure requires energy to break down. Thus the stronger better put together solid structures will require more energy to break apart. For alkanes, this can be seen from the graph above (i.e. the blue line).
The odd numbered alkanes have a lower trend in melting points that even numbered alkanes. This is because even numbered alkanes pack well in the solid phase, forming a well organised structure which requires more energy to break apart. The odd number alkanes pack less well and so the "looser" organised solid packing structure requires less energy to break apart. [14]
The melting points of branched-chain alkanes can be either higher or lower than those of the corresponding straight-chain alkanes, again this depends on the ability of the alkane in question to packing well in the solid phase: this is particularly true for isoalkanes (2-methyl isomers), which often have melting points higher than those of the linear analogues
Quote from wikipedia.org !
Conclusion:
The melting points increase less regularly regularly with molecular size because packing makes a difference in creating a solid, and the melting point will reflect packing efficiency as well as surface area
Regard !



